CERAMENT V is an injectable synthetic bone void filler consisting of 40% hydroxyapatite, 60% calcium sulfate and the antibiotic vancomycin hydrochloride.
The unique ratio of hydroxyapatite and calcium sulfate is designed to enable CERAMENT to resorb at the same rate that bone forms. Calcium sulfate acts as a resorbable carrier for hydroxyapatite, and hydroxyapatite is highly osteoconductive, promoting bone ingrowth.
The addition of vancomycin protects against colonization by vancomycin-sensitive microorganisms.
The characteristics of CERAMENT V mean that surgeons can manage bone defects in a more patient-friendly single-stage procedure. Moving to a single-stage from a multi-stage surgical protocol also frees healthcare resources to meet other needs, paving the way for a more cost-effective strategy for hospitals.
Remodels into host bone within 6-12 months
With CERAMENT, simultaneous de novo bone formation is seen throughout the material and it is fully remodeled into host bone within 6-12 months. Rapid bone remodeling reduces the risk of fracture, non-union and (re)infection
Injectible and flowable
CERAMENT can be injected through a 16G needle, making it ideal for minimally invasive procedures.
Flowability ensures excellent spread into and around a bone defect, for complete dead space management. This is particularly important for the healing of complex defects, where the extent of bone loss may not be apparent at first and certain areas may be difficult to access.
Vancomycin elution above MIC that is reliable and consistent
To protect the CERAMENT implant from bacterial colonization, it is essential to have a vancomycin concentration above MIC for a clinically relevant period of time.
In vitro studies have shown vancomycin elution from CERAMENT V has a high initial peak (>3000mg/L),
and remains above MIC for at least 28 days.
Studies of vancomycin concentrations in drain fluid from patients have confirmed that CERAMENT V mimics this elution pattern in vivo.
Irrespective of whether CERAMENT V is injected or formed into beads, the elution profile of vancomycin is the same.
The patented CERAMENT mixing and injecting device ensures a homogeneous distribution of antibiotic, whilst the material properties of CERAMENT mean that all of this antibiotic is made available for elution and delivered in a controlled fashion.
High local concentration of vancomycin, without high serum vancomycin levels
CERAMENT V has been designed to offer high local concentrations of vancomycin to effectively protect bone healing from vancomycin-sensitive organisms.
In vitro studies have shown that CERAMENT V offers vancomycin levels up to 3,000 times the minimum inhibitory concentration (MIC) for Methicillin-Resistant Staphylococcus aureus (MRSA).
In patients who underwent tumor resection arthroplasty with CERAMENT V implanted into the bone void, the mean blood plasma concentration of vancomycin measured was 0.3µg/mL (range 0.0 – 1.6µg/mL), well below the trough toxicity level of 10-20 µg/mL.
CERAMENT V
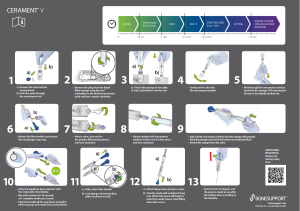
CERAMENT V Mixing Chart

CERAMENT V Product Fact Sheet
CERAMENT V – Value Guide

CERAMENT V Mixing Video

Instructions for use
Request previous version of the Instructions for Use here

Patient information

